 
The reactivity of group 1 elements increases as you go down the group because: the atoms become larger. The outermost electrons of the alkaline earth metals (group 2) are more difficult to remove than the outer electron of the alkali metals, leading to the group 2 metals being less reactive than those in group 1. As it requires less energy to form the ions, the reactivity increases. This is due to a decrease in ionisation energy as you progress down the group. Why does reactivity increase down Group 2?Īs you progress down Group 2, the reactivity increases. So down the group there is an increase in the atomic radius and hence the effective nuclear force decrease as the atomic radius increases and this leads to the decreased chemical reactivity down the group. 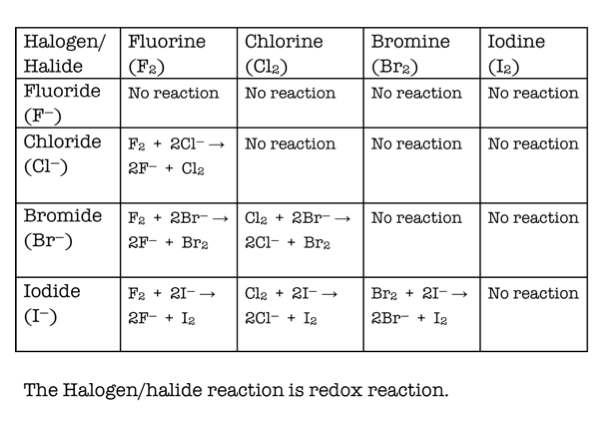
The chemical reactivity of group 17 elements decreases down the group. What happens to reactivity down the group 17? This lessens the attraction for valence electrons of other atoms, decreasing reactivity. This is due to the fact that atomic radius increases in size with an increase of electronic energy levels. The reactivities of the halogens decrease down the group ( At < I < Br < Cl < F). Why do halogens become less reactive down the group? the force of attraction between the nucleus and the outer electron decreases. the outer electron becomes further from the nucleus. Fluorine is the most reactive element of all in Group 7. This is the opposite trend to that seen in the alkali metals in Group 1 of the periodic table. The non-metal elements in Group 7 – known as the halogens – get less reactive as you go down the group. Do halogens get more reactive as you go down? the attraction between the nucleus and outer electron gets weaker as you go down the group – so the electron is more easily lost. 
The reactivity of Group 1 elements increases as you go down the group because: … the outer electron gets further from the nucleus as you go down the group. Why is Group 1 more reactive as you go down? As you move down the group, the amount of electron shielding increases, meaning that the electron is less attracted to the nucleus. This is because group 7 elements react by gaining an electron. the intermolecular forces become stronger. This is because, going down group 7: the molecules become larger. In group 7, the further down the group an element is, the higher its melting point and boiling point. Why do boiling points increase down group 7? This change manifests itself in a change in the phase of the elements from gas (F 2, Cl 2) to liquid (Br 2), to solid (I 2). The boiling points of halogens increase down the group due to the increasing strength of Van der Waals forces as the size and relative atomic mass of the atoms increase. What is the least reactive element in Group 6?.What happens to the reactivity of Group 0?.Why Fluorine is the most reactive element in Group 7?.Does reactivity increase down a period?.What is the least reactive group 7 element?.Why is iodine the least reactive halogen?.Why is Group 7 more reactive as you go up?.Does reactivity increase down a group 1?.Why does reactivity increase down Group 2?. 
What happens to reactivity down the group 17?.Why do halogens become less reactive down the group?.Do halogens get more reactive as you go down?.Why is Group 1 more reactive as you go down?.Why does reactivity decrease in group 7?.Why do boiling points increase down group 7?.What increases going down the halogen group?. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
